
Chemistry, 02.03.2020 17:06 aariannahnorwoo
At a particular temperature, K = 4.1 ✕ 10−6 for the following reaction. 2 CO2(g) 2 CO(g) + O2(g) If 2.3 moles of CO2 is initially placed into a 4.9-L vessel, calculate the equilibrium concentrations of all species.

Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 10:30
Which of the following characteristics are true of enzymes? check all that apply. a.)the structure of an enzyme can change if conditions change. b.)a single enzyme can normally catalyze a wide variety of reactions under many conditions. c.)enzymes are found only in nonliving systems. d.)enzymes allow living things to regulate body conditions through feedback mechanisms. e.)enzymes bind to specific substrates in specific ways. f.)enzymes increase the rate of reaction. g.)when shown in energy-reaction diagrams, enzymes represent the higher activation energy.
Answers: 1

Chemistry, 23.06.2019 16:40
Which heterogeneous mixture contains large particles that can settle out or can be filtered? colloid compound element suspension
Answers: 1

You know the right answer?
At a particular temperature, K = 4.1 ✕ 10−6 for the following reaction. 2 CO2(g) 2 CO(g) + O2(g) If...
Questions



Mathematics, 10.07.2019 20:20


English, 10.07.2019 20:20


Mathematics, 10.07.2019 20:20



English, 10.07.2019 20:20


Mathematics, 10.07.2019 20:20

Mathematics, 10.07.2019 20:20

Mathematics, 10.07.2019 20:20



Mathematics, 10.07.2019 20:30


Mathematics, 10.07.2019 20:30

![[O_2]](/tpl/images/0530/1146/b0db0.png) = 0.0124 = 12.4 ×10⁻³ M
= 0.0124 = 12.4 ×10⁻³ M![[CO]](/tpl/images/0530/1146/32558.png) = 0.0248 = 2.48 ×10⁻² M
= 0.0248 = 2.48 ×10⁻² M![[CO_2]](/tpl/images/0530/1146/5494d.png) = 0.4442 M
= 0.4442 M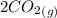 ⇄
⇄ 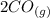 +
+ 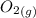
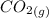 =
= 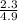 = 0.469
= 0.469![\frac{[CO]^2[O]}{[CO_2]^2}](/tpl/images/0530/1146/9940b.png)
![\frac{[2x]^2[x]}{[0.469-2x]^2}](/tpl/images/0530/1146/25de6.png)
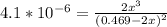
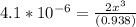
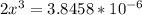
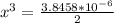
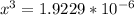
![x=\sqrt[3]{1.9929*10^{-6}}](/tpl/images/0530/1146/6ad64.png)



