Chemistry, 02.03.2020 16:51 vladisking888
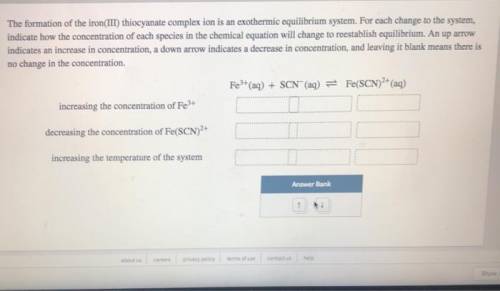
The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change to the system, indicate how the concentration of each species in the chemical equation will change to reestablish equilibrium. An up arrow indicates an increase in concentration, a down arrow indicates a decrease in concentration, and leaving it blank means there is no change in the concentration.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:30
The ph of carrots are 5.0 how it is classified a.acidic b.basic c.indicator d.neutral
Answers: 2

Chemistry, 21.06.2019 22:30
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1
You know the right answer?
The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each...
Questions



Mathematics, 12.09.2019 22:20

History, 12.09.2019 22:20


Social Studies, 12.09.2019 22:20


History, 12.09.2019 22:20





Mathematics, 12.09.2019 22:20

English, 12.09.2019 22:20

Mathematics, 12.09.2019 22:20


Biology, 12.09.2019 22:20


Mathematics, 12.09.2019 22:30

History, 12.09.2019 22:30