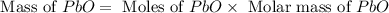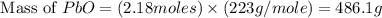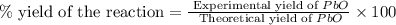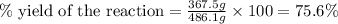Chemistry, 02.03.2020 16:32 StupidFatChipmunk
Consider the reaction. 2 Pb ( s ) + O 2 ( g ) ⟶ 2 PbO ( s ) An excess of oxygen reacts with 451.4 g of lead, forming 367.5 g of lead(II) oxide. Calculate the percent yield of the reaction.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
Consider the reaction. 2 Pb ( s ) + O 2 ( g ) ⟶ 2 PbO ( s ) An excess of oxygen reacts with 451.4 g...
Questions

Mathematics, 25.09.2019 04:00

Mathematics, 25.09.2019 04:00

Social Studies, 25.09.2019 04:00

Social Studies, 25.09.2019 04:00



Mathematics, 25.09.2019 04:00


Physics, 25.09.2019 04:00


Mathematics, 25.09.2019 04:00


Social Studies, 25.09.2019 04:00

Health, 25.09.2019 04:00

History, 25.09.2019 04:00



Biology, 25.09.2019 04:00


Biology, 25.09.2019 04:00

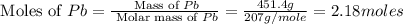
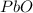
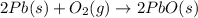
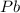 react to give 2 mole of
react to give 2 mole of