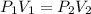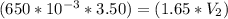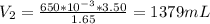Chemistry, 02.03.2020 04:06 letsgetcookingblog
A gas occupies a volume of 650.0 mL when the pressure is 3.50 atm. What will the new volume be if the pressure is reduced to 1.65 atm and the temperature remains constant.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 17:20
Which of these features are formed when hot groundwater is forced out through cracks in the earth's surface?
Answers: 2

Chemistry, 23.06.2019 06:00
Nthis lab, you will do experiments to identify types of changes. using the question format you learned (shown above), write an investigative question that you can answer by doing these experiments
Answers: 3
You know the right answer?
A gas occupies a volume of 650.0 mL when the pressure is 3.50 atm. What will the new volume be if th...
Questions




Mathematics, 03.05.2021 19:30


Social Studies, 03.05.2021 19:30


Mathematics, 03.05.2021 19:30


Mathematics, 03.05.2021 19:30

Chemistry, 03.05.2021 19:30

History, 03.05.2021 19:30


Mathematics, 03.05.2021 19:30


Mathematics, 03.05.2021 19:30


English, 03.05.2021 19:30

Mathematics, 03.05.2021 19:30