Chemistry, 29.02.2020 02:52 eriksprincess13
An aqueous solution was prepared at 21 oC by mixing 7.00 mL 2.00 x 10-2mol L-1Fe3+, 2.00 mL 1.50 x 10-3 mol L-1SCN−, and 1.00 mL water. At equilibrium, the concentration of the product complex, [Fe(SCN)2+]eq was determined to be 1.74 x 10-4mol L-1. What is the value of the equilibrium constant K for the reaction of interest at 21 oC?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

You know the right answer?
An aqueous solution was prepared at 21 oC by mixing 7.00 mL 2.00 x 10-2mol L-1Fe3+, 2.00 mL 1.50 x 1...
Questions



English, 05.10.2020 14:01

Social Studies, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01






English, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01


English, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01

English, 05.10.2020 14:01


English, 05.10.2020 14:01

 .....(1)
.....(1)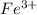 ions:
ions:

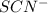 ions:
ions:
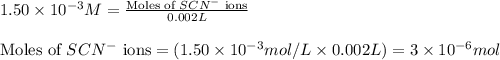


![[FeSCN^{2+}]](/tpl/images/0528/9422/797d4.png) complex follows:
complex follows:![Fe^{2+}+SCN^-\rightleftharpoons [FeSCN^{2+}]](/tpl/images/0528/9422/8a467.png)

 x
x![[FeSCN^{2+}]=1.74\times 10^{-4}M=x](/tpl/images/0528/9422/2c1de.png)
![[Fe^{2+}]\text{ ions}=(1.4\times 10^{-2}-x)=(1.4-0.0174)\times 10^{-3}=1.383\times 10^{-2}M](/tpl/images/0528/9422/f1fb2.png)
![[SCN^{-}]\text{ ions}=(3.0\times 10^{-4}-x)=(3.0-1.74)\times 10^{-4}=1.26\times 10^{-4}M](/tpl/images/0528/9422/6910c.png)
 for above equation follows:
for above equation follows:![K_{eq}=\frac{[FeSCN^{2+}]}{[Fe^{3+}][SCN^-]}](/tpl/images/0528/9422/63e7c.png)