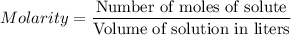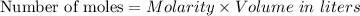Chemistry, 29.02.2020 01:25 samanthacruzsc51
For each trial, compute the mol of titrant; (molarity x L) and keep the number of significant figures to 4.
Trial 1: 12.49 mL =
mol NaOH
Trial 2: 12.32 mL =
mol NaOH
Trial 3: 11.87 mL =
mol NaOH

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 1


Chemistry, 23.06.2019 05:00
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2
You know the right answer?
For each trial, compute the mol of titrant; (molarity x L) and keep the number of significant figure...
Questions

Mathematics, 16.12.2020 20:10

English, 16.12.2020 20:10

Mathematics, 16.12.2020 20:10


Computers and Technology, 16.12.2020 20:10


Mathematics, 16.12.2020 20:10




Mathematics, 16.12.2020 20:10

Mathematics, 16.12.2020 20:10



Mathematics, 16.12.2020 20:10

Mathematics, 16.12.2020 20:10

Mathematics, 16.12.2020 20:10


Mathematics, 16.12.2020 20:10