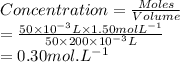Chemistry, 29.02.2020 00:08 2023greenlanden
A chemistry teacher adds 50.0 mL of 1.50 M H2SO4 solution to 200 mL of water. What is the concentration of the final solution?
Use M subscript i V subscript i equals M subscript f V subscript f..
0.300 M
0.375 M
6.00 M
7.50 M

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calcium hydroxide. if 26.5 ml of base are required to neutralize 20.3 ml of the acid, what is the molarity of the hydroiodic acid solution? m hydroiodic acid
Answers: 1



You know the right answer?
A chemistry teacher adds 50.0 mL of 1.50 M H2SO4 solution to 200 mL of water. What is the concentrat...
Questions

Mathematics, 24.05.2020 17:58

Biology, 24.05.2020 17:58

Engineering, 24.05.2020 17:58

Physics, 24.05.2020 17:58




Mathematics, 24.05.2020 17:58






Computers and Technology, 24.05.2020 17:58

Law, 24.05.2020 17:58

Mathematics, 24.05.2020 17:58


History, 24.05.2020 18:57


Social Studies, 24.05.2020 18:57

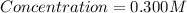
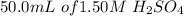 is added to 200ml of water.
is added to 200ml of water.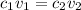 , where the latter relates to the final solution mix.
, where the latter relates to the final solution mix.