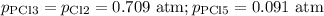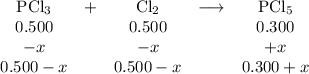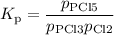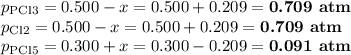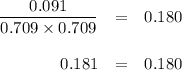Chemistry, 29.02.2020 00:17 Dogtes9667
For the exothermic reaction
PCl3(g)+Cl2(g)?PCl5(g)
Kp = 0.180 at a certain temperature.
A flask is charged with 0.500 atm PCl3 , 0.500 atm Cl2, and 0.300atm PCl5 at this temperature.
What are the equilibrium partial pressures of PCl3 , Cl2, and PCl5, respectively?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 01:30
How does the attraction between particles affect the ability of a solvent to dissolve in a substance
Answers: 1
You know the right answer?
For the exothermic reaction
PCl3(g)+Cl2(g)?PCl5(g)
Kp = 0.180 at a certain temperature.<...
PCl3(g)+Cl2(g)?PCl5(g)
Kp = 0.180 at a certain temperature.<...
Questions




Mathematics, 15.09.2021 23:30



Mathematics, 15.09.2021 23:30


Biology, 15.09.2021 23:30

English, 15.09.2021 23:30

Mathematics, 15.09.2021 23:30

World Languages, 15.09.2021 23:30


History, 15.09.2021 23:30

Mathematics, 15.09.2021 23:30



Computers and Technology, 15.09.2021 23:30

Advanced Placement (AP), 15.09.2021 23:30

Mathematics, 15.09.2021 23:30