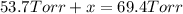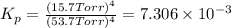Chemistry, 28.02.2020 22:46 AutumnJoy12
In a study of the reaction below at 1181 K, it was observed that when the equilibrium partial pressure of water vapor is 53.7 torr, the total pressure at equilibrium is 69.4 torr. Calculate Kp for this reaction at 1181 K. 3 Fe(s) + 4 H2O(g) equilibrium reaction arrow Fe3O4(s) + 4 H2(g)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2


Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1
You know the right answer?
In a study of the reaction below at 1181 K, it was observed that when the equilibrium partial pressu...
Questions






English, 22.06.2019 21:40


Mathematics, 22.06.2019 21:40


Arts, 22.06.2019 21:40




Mathematics, 22.06.2019 21:40


Mathematics, 22.06.2019 21:40

English, 22.06.2019 21:40

Mathematics, 22.06.2019 21:40


Computers and Technology, 22.06.2019 21:40

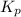 for this reaction at 1181 K is
for this reaction at 1181 K is  .
.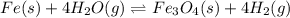
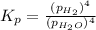
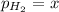
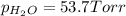
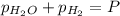 (Dalton's law of partial pressure)
(Dalton's law of partial pressure)