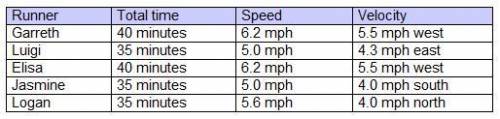
The chart indicates the time, speed, and velocity of five runners.
A. Garreth and Elisa r...

Chemistry, 28.02.2020 21:20 skyrileycasting
The chart indicates the time, speed, and velocity of five runners.
A. Garreth and Elisa ran the same distance but not the same displacement, Luigi and Jasmine went the same displacement, and Logan ran faster than Jasmine.
B. Garreth ran farther than Luigi, Elisa and Jasmine went the same distance, and Logan and Jasmine has the same displacement in the opposite direction.
C. Garreth and Elisa ran together, Luigi and Jasmine went the same distance, and Logan and Jasmine have the same displacement in the opposite direction.
D. Garreth ran less than Logan, Elisa and Jasmine went the same displacement, and Luigi and Garreth ran together.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
You know the right answer?
Questions

Chemistry, 01.12.2021 02:30

Computers and Technology, 01.12.2021 02:30

Computers and Technology, 01.12.2021 02:30

Mathematics, 01.12.2021 02:30

Chemistry, 01.12.2021 02:30




Chemistry, 01.12.2021 02:30



Mathematics, 01.12.2021 02:30




Social Studies, 01.12.2021 02:30






