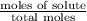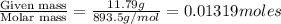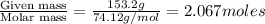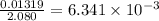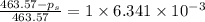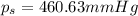Chemistry, 28.02.2020 19:30 samantha636
The vapor pressure of diethyl ether (ether) is 463.57 mm Hg at 25 °C. A nonvolatile, nonelectrolyte that dissolves in diethyl ether is chlorophyll. Calculate the vapor pressure of the solution at 25 °C when 11.79 grams of chlorophyll, C55H72MgN4O5 (893.5 g/mol), are dissolved in 153.2 grams of diethyl ether. diethyl ether = CH3CH2OCH2CH3 = 74.12 g/mol. VP(solution) = mm Hg

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

You know the right answer?
The vapor pressure of diethyl ether (ether) is 463.57 mm Hg at 25 °C. A nonvolatile, nonelectrolyte...
Questions

Advanced Placement (AP), 03.02.2020 05:01






Chemistry, 03.02.2020 05:01






Mathematics, 03.02.2020 05:01

Mathematics, 03.02.2020 05:01




Biology, 03.02.2020 05:01


Mathematics, 03.02.2020 05:01

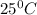 is 460.63 mmHg
is 460.63 mmHg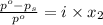
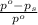 = relative lowering in vapor pressure
= relative lowering in vapor pressure
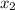 = mole fraction of solute =
= mole fraction of solute =