
Chemistry, 28.02.2020 18:56 kobiemajak
The value for Kw is 1.0 x 10-14 at 298K, but this value is temperature dependent. Given what you know about Kw and acid/base chemistry, what will the pH of water be at 283K if the value for Kw is 0.29 x 10-14? Is this solution acidic, basic, or neutral?
a. pH = 7.26, acidic
b. pH = 7.26, neutral
c. pH = 7.00, neutral
d. pH = 14.54, basic
e. pH = 7.26, basic

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:10
How does chemistry affect our world? a. chemicals makes our world more polluted. b. chemicals keeps us healthy. c. chemicals can or hurt our world. d. chemicals make our world safe to live in.
Answers: 1

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 23.06.2019 08:30
Plz a person walks 1 mile every day for exercise, leaving her front porch at 9 am and returning to her front porch at 9: 25 am what was the total displacement of her daily walk a. 1 mile b. 0 c. 25 min d. none of the above
Answers: 2

Chemistry, 23.06.2019 21:40
Hydrochloric acid reacts with barium hydroxide according to the equation: 2 hcl (aq) + ba(oh)2 (aq) → bacl2 (aq) + 2 h2o (l) δh = -118 kj calculate the heat (in kj) associated with the complete reaction of 109 grams of hcl (aq).
Answers: 3
You know the right answer?
The value for Kw is 1.0 x 10-14 at 298K, but this value is temperature dependent. Given what you kno...
Questions

Mathematics, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00


Mathematics, 13.12.2020 14:00


Biology, 13.12.2020 14:00


History, 13.12.2020 14:00

Physics, 13.12.2020 14:00

History, 13.12.2020 14:00


Mathematics, 13.12.2020 14:00

English, 13.12.2020 14:00

Computers and Technology, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00


Chemistry, 13.12.2020 14:00

English, 13.12.2020 14:00

English, 13.12.2020 14:00

Chemistry, 13.12.2020 14:00

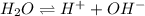
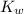
![K_w=[H^+][OH^-]](/tpl/images/0528/0368/bc68a.png)
![K_w=[H^+][H^+]=[H^+]^2](/tpl/images/0528/0368/7edc9.png)
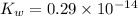
![K_w=[H^+]^2](/tpl/images/0528/0368/c7d3c.png)
![0.29\times 10^{-14}=[H^+]^2](/tpl/images/0528/0368/14418.png)
![[H^+]=5.385\times 10^{-8} M](/tpl/images/0528/0368/0b02c.png)
![pH=-\log [H^+]](/tpl/images/0528/0368/37e81.png)
![=-\log[5.385\times 10^{-8} M]=7.26](/tpl/images/0528/0368/66da4.png)


