
Chemistry, 28.02.2020 02:23 melissapulido198
Consider the equilibrium between SbCl5, SbCl3 and Cl2. SbCl5(g) -->SbCl3(g) + Cl2(g) K = 2.30×10-2 at 566 K .The reaction is allowed to reach equilibrium in a 7.40-L flask. At equilibrium, [SbCl5] = 0.333 M, [SbCl3] = 8.75×10-2 M and [Cl2] = 8.75×10-2 M.
(a) The equilibrium mixture is transferred to a 14.8-L flask. In which direction will the reaction proceed to reach equilibrium?
(b) Calculate the new equilibrium concentrations that result when the equilibrium mixture is transferred to a 14.8-L flask.
[SbCl5] = M
[SbCl3] = M
[Cl2] = M

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Y=‐1x + 7 if y has a value of ‐24 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1


Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1
You know the right answer?
Consider the equilibrium between SbCl5, SbCl3 and Cl2. SbCl5(g) -->SbCl3(g) + Cl2(g) K = 2.30×10-...
Questions

Physics, 08.11.2019 19:31

Mathematics, 08.11.2019 19:31

Mathematics, 08.11.2019 19:31


Mathematics, 08.11.2019 19:31

Mathematics, 08.11.2019 19:31







Biology, 08.11.2019 19:31






History, 08.11.2019 19:31

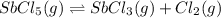
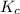
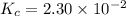
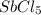 in 7.40 L = 0.333 M
in 7.40 L = 0.333 M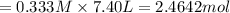
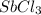 in 7.40 L =
in 7.40 L = 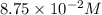
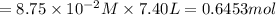
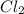 in 7.40 L =
in 7.40 L = 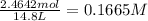
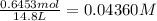
![K_c=\frac{[SbCl_3][Cl_2]}{[SbCl_5]}](/tpl/images/0527/6048/c5c78.png)
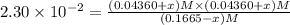
![[SbCl_5]=(0.1665-x) M=(0.1665-0.01536) M=0.1511 M](/tpl/images/0527/6048/35e3f.png)
![[SbCl_3]=(0.04360+x) M=(0.04360+0.01536) M=0.05896 M](/tpl/images/0527/6048/a8c15.png)
![[Cl_2]=(0.04360+x) M=(0.04360+0.01536) M=0.05896 M](/tpl/images/0527/6048/dab14.png)


