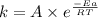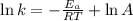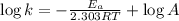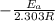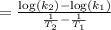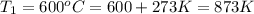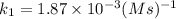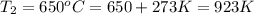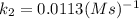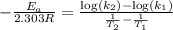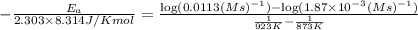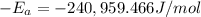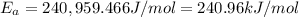The second-order rate constant for the decomposition of nitrous oxide to nitrogen molecules and oxygen atoms has been determined at various temperatures:
k(1/M·s) t(°C)
1.87 x10^-3 600
0.0113 650
0.0569 700
0.244 750
Determining the activation energy graphically.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1

Chemistry, 23.06.2019 02:00
Alice did an experiment to find the relationship between the angle at which a ray of light strikes a mirror and the angle at which the mirror reflects the light. she placed a ray box in front of a mirror. she changed the angle at which the light from the ray box struck the mirror and noted the corresponding angle at which the mirror reflected the light. which of the following is the dependent variable in this experiment? the mirror used to reflect the light the ray box used as the source of light angle at which the light from the ray box strikes the mirror angle at which the mirror reflects the light from the ray box
Answers: 2

Chemistry, 23.06.2019 05:30
Elizabeth has two separate samples of the same substance. sample is in the liquid state, and the other is in the solid state. the two samples most likely differ in which property?
Answers: 1
You know the right answer?
The second-order rate constant for the decomposition of nitrous oxide to nitrogen molecules and oxyg...
Questions



SAT, 31.12.2019 01:31


SAT, 31.12.2019 01:31

Mathematics, 31.12.2019 01:31



Mathematics, 31.12.2019 01:31

Biology, 31.12.2019 01:31



History, 31.12.2019 01:31

Social Studies, 31.12.2019 01:31

Mathematics, 31.12.2019 01:31


Mathematics, 31.12.2019 01:31

History, 31.12.2019 01:31

History, 31.12.2019 01:31

Mathematics, 31.12.2019 01:31