
Chemistry, 27.02.2020 05:24 arianaguerin
What mass of NH3 (in grams) must be used to produce 5.65 tons of HNO3 by the Ostwald process, assuming an 80.0 percent yield in each step (1 ton = 2000 lb; 1 lb = 453.6 g)? Enter your answer in scientific notation.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2


Chemistry, 23.06.2019 07:20
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3
You know the right answer?
What mass of NH3 (in grams) must be used to produce 5.65 tons of HNO3 by the Ostwald process, assumi...
Questions



English, 29.09.2021 23:10



History, 29.09.2021 23:10

History, 29.09.2021 23:10



History, 29.09.2021 23:10

English, 29.09.2021 23:10

History, 29.09.2021 23:10



Mathematics, 29.09.2021 23:10



Mathematics, 29.09.2021 23:10

English, 29.09.2021 23:10

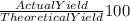 = 80 %
= 80 %

