

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1


Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

You know the right answer?
7. A 28.4 g sample of aluminum is heated to 39.4 °C, then is placed in a calorimeter
con...
con...
Questions


English, 31.03.2020 23:29

Mathematics, 31.03.2020 23:29


English, 31.03.2020 23:29


Mathematics, 31.03.2020 23:29


Biology, 31.03.2020 23:29

English, 31.03.2020 23:29

Mathematics, 31.03.2020 23:29

History, 31.03.2020 23:29



Mathematics, 31.03.2020 23:30

English, 31.03.2020 23:30



Business, 31.03.2020 23:30

Biology, 31.03.2020 23:30

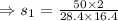
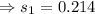 calorie /gram °C
calorie /gram °C

