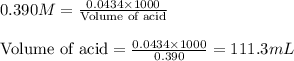Chemistry, 27.02.2020 02:21 jaidalynkimora
Suppose that you have 115 mL of a buffer that is 0.460 M in both benzoic acid ( C 6 H 5 COOH ) and its conjugate base ( C 6 H 5 COO − ) . Calculate the maximum volume of 0.390 M HCl that can be added to the buffer before its buffering capacity is lost.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
One mole of zinc has a mass of 65.4 grams. approximately how many atoms of zinc are present in one mole of zinc? 32 × 1023 atoms 6 × 1023 atoms 66 atoms 65 atoms
Answers: 1

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
Suppose that you have 115 mL of a buffer that is 0.460 M in both benzoic acid ( C 6 H 5 COOH ) and i...
Questions


Social Studies, 27.08.2019 20:10


Mathematics, 27.08.2019 20:10

English, 27.08.2019 20:10

Mathematics, 27.08.2019 20:10



History, 27.08.2019 20:10



English, 27.08.2019 20:10






Mathematics, 27.08.2019 20:10

Computers and Technology, 27.08.2019 20:10

![pH=pK_a+\log(\frac{[salt]}{[acid]})](/tpl/images/0526/1364/e4eea.png)
![pH=pK_a+\log(\frac{[C_6H_5COO^-]}{[C_6H_5COOH]})](/tpl/images/0526/1364/e41e3.png) .....(1)
.....(1)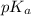 = negative logarithm of acid dissociation constant of benzoic acid = 4.2
= negative logarithm of acid dissociation constant of benzoic acid = 4.2![[C_6H_5COOH]=0.460M](/tpl/images/0526/1364/fc044.png)
![[C_6H_5COO^-]=0.460M](/tpl/images/0526/1364/ef922.png)
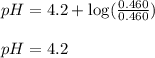
![3.2=4.2+\log(\frac{[C_6H_5COO^-]}{[C_6H_5COOH]})\\\\\frac{[C_6H_5COO^-]}{[C_6H_5COOH]}=0.1](/tpl/images/0526/1364/89428.png)
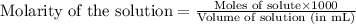 ......(2)
......(2)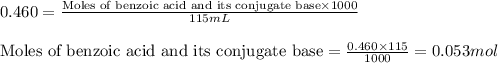
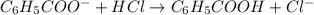
![\frac{[C_6H_5COO^-]-x}{[C_6H_5COOH]+x}=0.1\\\\\frac{0.053-x}{0.053+x}=0.1\\\\x=0.0434](/tpl/images/0526/1364/881c9.png)