
Chemistry, 27.02.2020 02:02 TombRaider167
Deep-sea divers must use special gas mixtures in their tanks, rather than compressed air, to avoid serious problems. One such breathing mixture contains helium, oxygen, and carbon dioxide. Determine the partial pressure of oxygen when the total pressure in the tank is 201.4 kPa if PHe = 125.4 kPa and PCO2= 18.2 kPa? Must show all work that leads to answer for credit

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1


Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1
You know the right answer?
Deep-sea divers must use special gas mixtures in their tanks, rather than compressed air, to avoid s...
Questions





Chemistry, 28.02.2021 21:10

Mathematics, 28.02.2021 21:10


Mathematics, 28.02.2021 21:10


Biology, 28.02.2021 21:10

Physics, 28.02.2021 21:10




English, 28.02.2021 21:10

Mathematics, 28.02.2021 21:10

Mathematics, 28.02.2021 21:10

Medicine, 28.02.2021 21:10

Mathematics, 28.02.2021 21:10

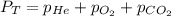
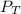 = 201.4 kPa
= 201.4 kPa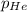 = 125.4 kPa
= 125.4 kPa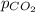 = 18.2 kPa
= 18.2 kPa![201.4=125.4+p_{O_2}+18.2\\\\p_{O_2}=201.4-[125.4+18.2]=57.8kPa](/tpl/images/0526/1098/fa869.png)


