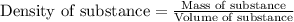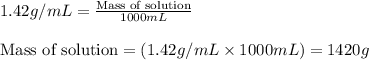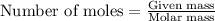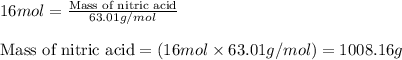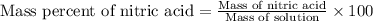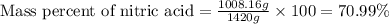Chemistry, 26.02.2020 23:49 garrettrhoad
Ommercially available aqueous nitric acid has a density of 1.42 g/mL and a concentration of 16 M. Calculate the mass percent of HNO3 (molar mass = 63.01 g/mol) in the solution.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:50
Which statement describes how phase changes can be diagrammed as a substance is heated? the phase is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the phase is on the x-axis. the time is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the time is on the x-axis.
Answers: 1

Chemistry, 22.06.2019 01:40
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3

Chemistry, 22.06.2019 08:00
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1
You know the right answer?
Ommercially available aqueous nitric acid has a density of 1.42 g/mL and a concentration of 16 M. Ca...
Questions




Mathematics, 11.10.2019 10:30

Mathematics, 11.10.2019 10:30


Mathematics, 11.10.2019 10:30


Mathematics, 11.10.2019 10:30

Biology, 11.10.2019 10:30


Biology, 11.10.2019 10:30

English, 11.10.2019 10:30

Geography, 11.10.2019 10:30


Biology, 11.10.2019 10:30

Mathematics, 11.10.2019 10:30


Health, 11.10.2019 10:30

Mathematics, 11.10.2019 10:30