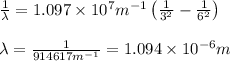Chemistry, 26.02.2020 23:22 pennygillbert
Calculate the wavelength of the photon emitted when an electron makes a transition from n=6 to n=3. You can make use of the following constants: h=6.626×10−34 J⋅s c=2.998×108 m/s 1 m=109 nm

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 22:30
Each of the following compounds contains a metal that can exhibit more than one ionic charge. provide systematic names for each of these compounds. (a) cr(clo3)6 (b) mo(cn)6 (c) cr2(so3)3 (d) v(clo2)2 (e) v(cn)5 (f) os(clo2)4
Answers: 3

Chemistry, 22.06.2019 13:40
Can someone me with 6 to 10 plz this is for masteries test.
Answers: 1

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2
You know the right answer?
Calculate the wavelength of the photon emitted when an electron makes a transition from n=6 to n=3....
Questions

History, 04.07.2019 23:00

Mathematics, 04.07.2019 23:00




History, 04.07.2019 23:00

Mathematics, 04.07.2019 23:00

Mathematics, 04.07.2019 23:00

History, 04.07.2019 23:00

Biology, 04.07.2019 23:00


Biology, 04.07.2019 23:00





History, 04.07.2019 23:00

English, 04.07.2019 23:00


Mathematics, 04.07.2019 23:00

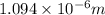
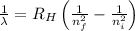
 = Wavelength of radiation
= Wavelength of radiation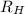 = Rydberg's Constant =
= Rydberg's Constant = 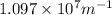
 = Final energy level = 3
= Final energy level = 3 = Initial energy level = 6
= Initial energy level = 6