
A voltaic cell is set up with copper and hydrogen half-cells. Standard conditions are used in the copper half-cell, Cu2+ (aq, 1.00 M) | Cu (s). The hydrogen gas pressure is 1.00 bar. A value of 0.490 V is recorded for E Cell at 298 K. Determine the concentration of H+ and the pH of the solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2

Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1

Chemistry, 23.06.2019 02:20
Why dose heating increase the speed at which a solution dissolved in water
Answers: 1
You know the right answer?
A voltaic cell is set up with copper and hydrogen half-cells. Standard conditions are used in the co...
Questions


Mathematics, 01.07.2019 22:30



Chemistry, 01.07.2019 22:30


Mathematics, 01.07.2019 22:30

Mathematics, 01.07.2019 22:30




Biology, 01.07.2019 22:30


Social Studies, 01.07.2019 22:30

Mathematics, 01.07.2019 22:30


English, 01.07.2019 22:30

History, 01.07.2019 22:30

Mathematics, 01.07.2019 22:30

Social Studies, 01.07.2019 22:30

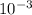 M and the pH = 2.6 of the solution
M and the pH = 2.6 of the solution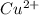 ) is the cathode and hydrogen (
) is the cathode and hydrogen (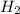 ) is the anode.
) is the anode.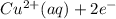 ⇒ Cu(s)
⇒ Cu(s)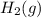 ⇒
⇒ 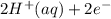
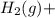
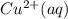 ⇒
⇒ 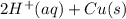
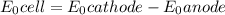
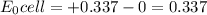
![\frac{[H^{+}]^{2} }{[Cu^{2+}]P_{H2} }](/tpl/images/0525/6315/22043.png)
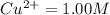 but
but 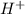 is unknown. we solve this using hernst equation.
is unknown. we solve this using hernst equation.![E = E^{0} -\frac{0.0257}{n}ln\frac{[H^{+}]^{2} }{[Cu^{2+}]P_{H2} }](/tpl/images/0525/6315/8e268.png)
![0.490 = 0.337 -\frac{0.0257}{2}ln\frac{[H^{+}]^{2} }{[1][1]}](/tpl/images/0525/6315/43505.png)
![ln{[H^{+}]^{2} } = -11.9](/tpl/images/0525/6315/8c81e.png)
![2ln{[H^{+}] } = -11.9](/tpl/images/0525/6315/31a40.png)
![ln{[H^{+}] } = -5.95](/tpl/images/0525/6315/e2ee5.png)
![[H^{+}] = 3* 10^{-3} M](/tpl/images/0525/6315/d9bb9.png)


