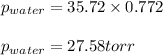Chemistry, 26.02.2020 20:52 cocomorillo35181
The ambient temperature is 85.0°F and the humidity of the surrounding air is reported to be 68.0%. Using the Clausius-Clapeyron equation and the boiling point of water as 100.0°C at 760 torr, calculate the vapor pressure (in torr) of water in the air. Use 40.7 kJ/mol as the ∆Hvap of water.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1
You know the right answer?
The ambient temperature is 85.0°F and the humidity of the surrounding air is reported to be 68.0%. U...
Questions

Mathematics, 02.02.2021 17:40

English, 02.02.2021 17:40






English, 02.02.2021 17:40



SAT, 02.02.2021 17:40







Chemistry, 02.02.2021 17:40


Mathematics, 02.02.2021 17:40

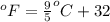
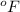 = temperature in Fahrenheit
= temperature in Fahrenheit 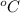 = temperature in centigrade
= temperature in centigrade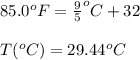
![\ln(\frac{P_2}{P_1})=\frac{\Delta H}{R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0525/3984/5c76e.png)
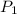 = initial pressure which is the pressure at normal boiling point = 760 torr
= initial pressure which is the pressure at normal boiling point = 760 torr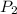 = final pressure = ?
= final pressure = ?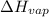 = Enthalpy of vaporization = 40.7 kJ/mol = 40700 J/mol (Conversion factor: 1 kJ = 1000 J)
= Enthalpy of vaporization = 40.7 kJ/mol = 40700 J/mol (Conversion factor: 1 kJ = 1000 J)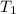 = initial temperature =
= initial temperature = ![100^oC=[100+273]K=373K](/tpl/images/0525/3984/44e24.png)
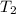 = final temperature =
= final temperature = ![29.44^oC=[29.44+273]=302.44K](/tpl/images/0525/3984/ddd83.png)
![\ln(\frac{P_2}{760})=\frac{40700J/mol}{8.314J/mol.K}[\frac{1}{373}-\frac{1}{302.44}]\\\\P_2=35.72torr](/tpl/images/0525/3984/e0926.png)
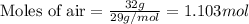
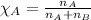

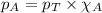
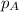 = vapor pressure of water = ?
= vapor pressure of water = ?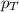 = total pressure = 35.72 torr
= total pressure = 35.72 torr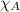 = mole fraction of water = 0.774
= mole fraction of water = 0.774