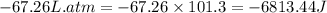Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Aroller coaster car is traveling down a track at 22 m/s. the car has a mass of 2000 kg. what is the kinetic energy of the car? a) 22,000 j b) 968,000 j c) 484,000 j d) 44,000 j
Answers: 2

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1
You know the right answer?
The combustion of 0.25 moles of octane gas (C8H18) to CO2 gas and H2O gas (vapor) against a constant...
Questions


Mathematics, 29.12.2019 17:31

English, 29.12.2019 17:31





History, 29.12.2019 17:31





Biology, 29.12.2019 17:31

World Languages, 29.12.2019 17:31

Mathematics, 29.12.2019 17:31



History, 29.12.2019 17:31

Mathematics, 29.12.2019 17:31

Mathematics, 29.12.2019 17:31


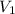 = initial volume = 113.1 L
= initial volume = 113.1 L = final volume = 148.5 L
= final volume = 148.5 L