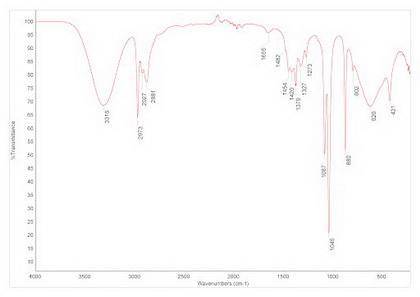
In a fischer esterification, a carboxylic acid and an alcohol combine in the presence of acid to make an ester and a molecule of water. the infrared spectrum shown below represents the substance isolated at the end of the reaction. use the infrared spectrum to identify the substance, and infer the success of the reaction.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

You know the right answer?
In a fischer esterification, a carboxylic acid and an alcohol combine in the presence of acid to mak...
Questions

Social Studies, 28.01.2021 06:10

Mathematics, 28.01.2021 06:10

Mathematics, 28.01.2021 06:10



Mathematics, 28.01.2021 06:10

Mathematics, 28.01.2021 06:10

Mathematics, 28.01.2021 06:10

Biology, 28.01.2021 06:10

Mathematics, 28.01.2021 06:10

Computers and Technology, 28.01.2021 06:10

Social Studies, 28.01.2021 06:10




Mathematics, 28.01.2021 06:10