Consider the following elementary reaction:
N2O—> N2 + O
suppose we let K1 stand fo...

Consider the following elementary reaction:
N2O—> N2 + O
suppose we let K1 stand for the rate constant of this reaction, and k-1 stand for the rate constant of the reverse reaction. write an expression that gives the equilibrium concentration of N2O in terms of k1, k-1 and the equilibrium concentrations of N2 and O

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:50
Which real-world scenarios below represent physical and chemical changes? -running a car -exploding fireworks -mixing water and powdered drink mix -combining oil and vinegar to make salad dressing -taking aspirin for a headache -diluting bleach with water-digesting dinner-spreading peanut butter on bread
Answers: 2

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3
You know the right answer?
Questions




Biology, 12.01.2020 04:31

English, 12.01.2020 04:31

Biology, 12.01.2020 04:31




Mathematics, 12.01.2020 04:31


Mathematics, 12.01.2020 04:31

Spanish, 12.01.2020 04:31

History, 12.01.2020 04:31


Mathematics, 12.01.2020 04:31

History, 12.01.2020 04:31

History, 12.01.2020 04:31


Biology, 12.01.2020 04:31

![[N_2O]=\frac{K^{-1}}{K_1}\times [N_2][O_2]](/tpl/images/0524/0986/7a4ba.png)
![R_f=K_1\times [N_2O]](/tpl/images/0524/0986/5d806.png)
![R_b=K^{-1}\times [N_2][O_2]](/tpl/images/0524/0986/327e3.png)
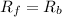
![K_1\times [N_2O]=K^{-1}\times [N_2][O_2]](/tpl/images/0524/0986/21d13.png)


