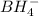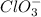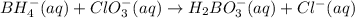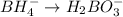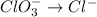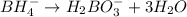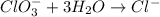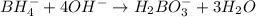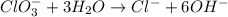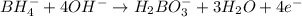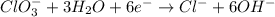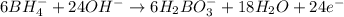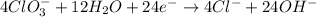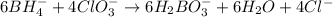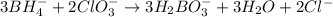Balance the following skeleton reaction and identify the oxidizing and reducing agents: Include the states of all reactants and products in your balanced equation. You do not need to include the states with the identities of the oxidizing and reducing agents.
BH4−(aq) + ClO3−(aq) → H2BO3−(aq) + Cl−(aq) [basic]
a. The oxidizing agent is:
b. The reducing agent is:

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1


Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1
You know the right answer?
Balance the following skeleton reaction and identify the oxidizing and reducing agents: Include the...
Questions

Mathematics, 09.12.2021 01:00

Social Studies, 09.12.2021 01:00

English, 09.12.2021 01:00


Mathematics, 09.12.2021 01:00

Social Studies, 09.12.2021 01:00



Mathematics, 09.12.2021 01:00



Mathematics, 09.12.2021 01:00








Social Studies, 09.12.2021 01:00