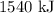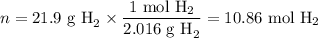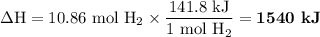Chemistry, 25.02.2020 22:09 lucyamine0
When 1 mole of hydrogen gas (H2) reacts with excess oxygen to form water at a constant pressure, 241.8 KJ of energy is released as heat. Calculate ΔH for a process in which 21.9 g sample of hydrogen gas (H2) reacts with excess oxygen at constant pressure.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2


You know the right answer?
When 1 mole of hydrogen gas (H2) reacts with excess oxygen to form water at a constant pressure, 241...
Questions

Mathematics, 21.10.2019 15:50



Mathematics, 21.10.2019 15:50





History, 21.10.2019 15:50







Mathematics, 21.10.2019 15:50

Social Studies, 21.10.2019 15:50



Mathematics, 21.10.2019 15:50