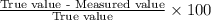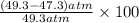Chemistry, 25.02.2020 21:59 sierram298
In Part B the given conditions were 1.00 mol of argon in a 0.500-L container at 27.0 ∘C . You identified that the ideal pressure (Pideal) was 49.3 atm , and the real pressure (Preal) was 47.3 atm under these conditions. The percent difference between the ideal and real gas is .

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3
You know the right answer?
In Part B the given conditions were 1.00 mol of argon in a 0.500-L container at 27.0 ∘C . You identi...
Questions

Spanish, 13.10.2020 21:01



Mathematics, 13.10.2020 21:01

Computers and Technology, 13.10.2020 21:01

Arts, 13.10.2020 21:01





Mathematics, 13.10.2020 21:01








Mathematics, 13.10.2020 21:01