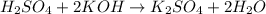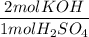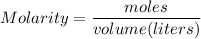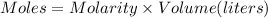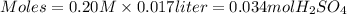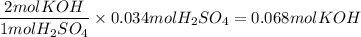The reaction of sulfuric acid (H2SO4) with potassium hydroxide (KOH) is described by the equation below. Suppose
0.06 L of KOH with unknown concentration is placed in a flask with bromthymol blue indicator. A solution of 0.20 M
H2504 is dripped into the KOH solution. After exactly 0.017 L of H2SO4 is added, the Indicator changes from blue to
yellow. What is the concentration of the KOH? You must show all of your work to earn credit. (4 points)
H2SO4 + 2KOH → K2SO4 + 2H20

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 02:00
Alice did an experiment to find the relationship between the angle at which a ray of light strikes a mirror and the angle at which the mirror reflects the light. she placed a ray box in front of a mirror. she changed the angle at which the light from the ray box struck the mirror and noted the corresponding angle at which the mirror reflected the light. which of the following is the dependent variable in this experiment? the mirror used to reflect the light the ray box used as the source of light angle at which the light from the ray box strikes the mirror angle at which the mirror reflects the light from the ray box
Answers: 2

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1

Chemistry, 23.06.2019 02:00
What are fossils of organisms that existed over a wide area but only for a limited time period called?
Answers: 2

You know the right answer?
The reaction of sulfuric acid (H2SO4) with potassium hydroxide (KOH) is described by the equation be...
Questions


Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Chemistry, 11.02.2021 01:00

Chemistry, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00



History, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00




Mathematics, 11.02.2021 01:00

Social Studies, 11.02.2021 01:00