
A compound containing chromium, Cr; chlorine, Cl; and oxygen, O, is analyzed and found to be 33.6% chromium, 45.8% chlorine, and 20.6% oxygen by mass. What is the empirical formula of the compound? The molar mass of chromium, Cr, is 51.996 gmol; the molar mass of chlorine, Cl, is 35.45 gmol; and the molar mass of oxygen, O, is 15.999 gmol.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:40
Astudent made the lewis dot diagram of a compound shown. what is the error in the lewis dot diagram? a)an o atom should transfer all of its six electrons to mg because the formula is mgo b) both electrons of mg should be transferred to one o adam because the formula is mgo c) the electrons should be transferred from each o add him to capital mg because mg has fewer electrons d) the number of dots around mg should be four because it has to transfer two electrons to each o
Answers: 1

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1
You know the right answer?
A compound containing chromium, Cr; chlorine, Cl; and oxygen, O, is analyzed and found to be 33.6% c...
Questions

Chemistry, 18.06.2020 19:57






Mathematics, 18.06.2020 19:57







Computers and Technology, 18.06.2020 19:57


Mathematics, 18.06.2020 19:57


Mathematics, 18.06.2020 19:57

Mathematics, 18.06.2020 19:57

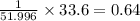 mol
mol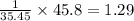 mol.
mol.
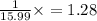 mol.
mol.
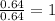
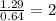
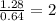
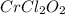
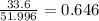
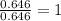 1
1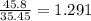
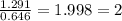 2
2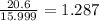
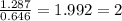 2
2



