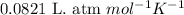Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
How many moles of O2 gas are there in a 1.0 L container which contains 0.12 atm of O2 gas at 25 ºC?...
Questions

Chemistry, 26.02.2021 01:10



Mathematics, 26.02.2021 01:10




Advanced Placement (AP), 26.02.2021 01:10


Mathematics, 26.02.2021 01:10



Advanced Placement (AP), 26.02.2021 01:10

Mathematics, 26.02.2021 01:10


Computers and Technology, 26.02.2021 01:10




Mathematics, 26.02.2021 01:10

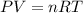
![25^oC=[25+273]K=298K](/tpl/images/0522/8031/df1f6.png)