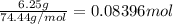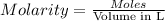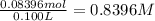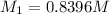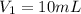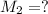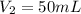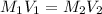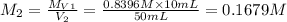Chemistry, 25.02.2020 04:01 swelch2010
. A student transferred a 10-mL aliquot of a stock sodium hypochlorite solution into a 50-mL volumetric flask using a volumetric pipet. The solution was then diluted to the mark with distilled water. Assuming that the concentration of a stock sodium hypochlorite is 6.25% (w/v), calculate the molarity of the diluted sodium hypochlorite solution. Molar mass of sodium hypochlorite

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
. A student transferred a 10-mL aliquot of a stock sodium hypochlorite solution into a 50-mL volumet...
Questions


English, 31.01.2020 14:46

Chemistry, 31.01.2020 14:46

Mathematics, 31.01.2020 14:46

Mathematics, 31.01.2020 14:46


Mathematics, 31.01.2020 14:46

Mathematics, 31.01.2020 14:46

English, 31.01.2020 14:46


Geography, 31.01.2020 14:46


Biology, 31.01.2020 14:46




Biology, 31.01.2020 14:46

Mathematics, 31.01.2020 14:46

History, 31.01.2020 14:46