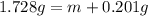Chemistry, 25.02.2020 03:43 ninaaforever
1. Assuming that you recovered 100% of the acetanilide in the sample, how much of the mass of the original sample was made up of impurities?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2


Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2
You know the right answer?
1. Assuming that you recovered 100% of the acetanilide in the sample, how much of the mass of the or...
Questions

Mathematics, 24.03.2020 20:18



Mathematics, 24.03.2020 20:18

Mathematics, 24.03.2020 20:18





Computers and Technology, 24.03.2020 20:18

Mathematics, 24.03.2020 20:18

Social Studies, 24.03.2020 20:18

Biology, 24.03.2020 20:18

History, 24.03.2020 20:18



Mathematics, 24.03.2020 20:18



Mathematics, 24.03.2020 20:18