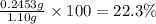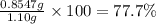Chemistry, 25.02.2020 03:23 alizeleach0123
>A 1.10-g sample contains only glucose (C6H12O6) and sucrose (C12H22O11). When a sample is dissolved in water to a total solution volume of 25.0 mL, the osmotic pressure of the solution is 3.78 atm at 298 K. What is the percent mass of the glucose in the sample, and what is the percent mass of sucrose in the sample?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 00:30
There are approximately 15 milliliters (ml) in 1 tablespoon (tbsp). what expression can be used to find the approximate number of milliliters in 3 tbsp?
Answers: 1
You know the right answer?
>A 1.10-g sample contains only glucose (C6H12O6) and sucrose (C12H22O11). When a sample is dissol...
Questions

Social Studies, 13.07.2019 13:00








Social Studies, 13.07.2019 13:00







Mathematics, 13.07.2019 13:00

Biology, 13.07.2019 13:00



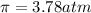
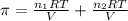
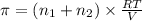
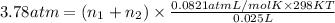
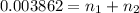
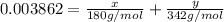
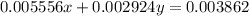 ..[2]
..[2]