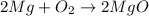Chemistry, 25.02.2020 03:04 tonimgreen17p6vqjq
A student burned 0.250 g of magnesium metal in an open crucible. Oxygen from the air combined with the metal forming magnesium oxide. If 0.421 g of magnesium oxide were produced, what mass of oxygen combined with the metal? 1. 0.171 g 2. 0.671 g 3. 1.684 g 4. 0.594 g

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Order the following from smallest to largest atom, electron, quark, proton, neutron, molecule, nucleus
Answers: 1

Chemistry, 21.06.2019 17:50
Which statement is a reason to support population regulation? a) it is unethical for us to control birth control rates b) humans have the freedom to produce as many children as desired c) the gap between the rich and poor has been narrowing since 1960 d) billions more people on the earth will intensify many environmental and social problems
Answers: 1

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3
You know the right answer?
A student burned 0.250 g of magnesium metal in an open crucible. Oxygen from the air combined with t...
Questions

Mathematics, 12.12.2019 00:31

History, 12.12.2019 00:31




History, 12.12.2019 00:31





Mathematics, 12.12.2019 00:31




Social Studies, 12.12.2019 00:31


Social Studies, 12.12.2019 00:31

Mathematics, 12.12.2019 00:31

Mathematics, 12.12.2019 00:31

English, 12.12.2019 00:31