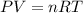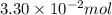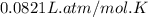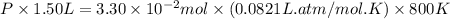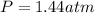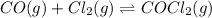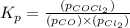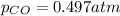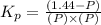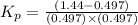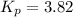Chemistry, 24.02.2020 22:28 pattydixon6
Pure phosgene gas (COCl2,), 3.30 x 10-2 mol, was placed in a 1.50-L container. It was heated to 800K, and at equilibrium the pressure of CO was found to be 0.497 atm. Calculate the equilibrium constant, Kp, for the reaction: CO(g) Cl2(g) COCl2(g)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
This is a mixture that has the same composition throughout.
Answers: 1

Chemistry, 22.06.2019 01:00
Which of the following is always a reactant in a combustion reaction? oxygen nitrogen hydrogen carbon
Answers: 1

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1

Chemistry, 23.06.2019 06:30
Which of these natural resources is non-renewable a.corn b.wind c.geothermal d.natural gas
Answers: 2
You know the right answer?
Pure phosgene gas (COCl2,), 3.30 x 10-2 mol, was placed in a 1.50-L container. It was heated to 800K...
Questions

SAT, 09.12.2021 02:40

Mathematics, 09.12.2021 02:40

SAT, 09.12.2021 02:40



Social Studies, 09.12.2021 02:40





Biology, 09.12.2021 02:40



Social Studies, 09.12.2021 02:40


Computers and Technology, 09.12.2021 02:40




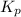 for the reaction is, 3.82
for the reaction is, 3.82