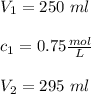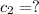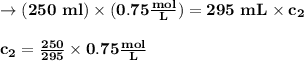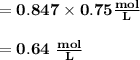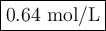Chemistry, 23.02.2020 04:47 brainlord4209
Dilutions Worksheet - Solutions
If 45 mL of water are added to 250 mL of a 0.75 M K2SO4 solution, what will the
molarity of the diluted solution be?
(0.75 M)(250 ml) = M2 (295 mL)
M2 = (0.75 M) (250 mL) = 0.64 M
(295 mL)
Where did the 295ml came from

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2
You know the right answer?
Dilutions Worksheet - Solutions
If 45 mL of water are added to 250 mL of a 0.75 M K2SO4 soluti...
If 45 mL of water are added to 250 mL of a 0.75 M K2SO4 soluti...
Questions




Engineering, 23.08.2019 04:10




Social Studies, 23.08.2019 04:10






Engineering, 23.08.2019 04:10


Mathematics, 23.08.2019 04:10


Computers and Technology, 23.08.2019 04:10


Computers and Technology, 23.08.2019 04:10