
Chemistry, 22.02.2020 03:32 bibigardeax
Mastering chem If the experiment below is run for 60 s, 0.16 mol A remain. Which of the following statements is or are true? At time 0 seconds, a flask contains 1.00 mole of A and 0 moles of B. At 20 seconds, the flask holds 0.54 moles of A and 0.46 moles of B. At 40 seconds, the flask holds 0.3 moles of A and 0.7 moles of B. After 60 s there are 0.84 mol B in the flask. The decrease in the number of moles of A from t1 = 0 to t2 = 20 s is greater than that for t1 = 40 to t2 = 60 s. The average rate for the reaction from t1 = 40 to t2 = 60 s is 7.0×10−3 M/s.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 03:00
Schrodinger and heisenberg developed an alternate theory about atomic nature that contradicted some of bohr's model of the atom. how do changes resulting from new technology and evidence affect the reputation of the atomic theory?
Answers: 1

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1
You know the right answer?
Mastering chem If the experiment below is run for 60 s, 0.16 mol A remain. Which of the following st...
Questions

Mathematics, 06.07.2019 01:30


Physics, 06.07.2019 01:30

Mathematics, 06.07.2019 01:30




Mathematics, 06.07.2019 01:30



Mathematics, 06.07.2019 01:30

History, 06.07.2019 01:30


Mathematics, 06.07.2019 01:30



English, 06.07.2019 01:40


Spanish, 06.07.2019 01:40

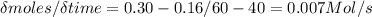 which is true as well
which is true as well 


