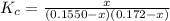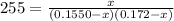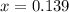Chemistry, 21.02.2020 21:58 leahstubbs
For the following reaction, Kc = 255 at 1000 K. CO(g)+Cl2(g)⇌COCl2(g) A reaction mixture initially contains a CO concentration of 0.1550 M and a Cl2 concentration of 0.172 M at 1000 K.
What is the equilibrium concentration of CO at 1000 K?
What is the equilibrium concentration of Cl2 at 1000 K?
What is the equilibrium concentration of COCl2 at 1000 K?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which substances have the lowest melting points: ionic covalent, or metallic
Answers: 1


Chemistry, 22.06.2019 23:00
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1

Chemistry, 23.06.2019 03:40
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.
Answers: 2
You know the right answer?
For the following reaction, Kc = 255 at 1000 K. CO(g)+Cl2(g)⇌COCl2(g) A reaction mixture initially c...
Questions




Mathematics, 02.10.2020 15:01



History, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Chemistry, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01




Mathematics, 02.10.2020 15:01

History, 02.10.2020 15:01

Biology, 02.10.2020 15:01

Medicine, 02.10.2020 15:01

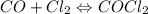
 initial
initial![K_c = \frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0519/6554/f9140.png)
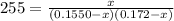
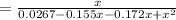
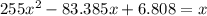
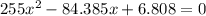
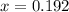
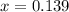 (correct value)
(correct value)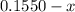
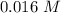
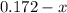
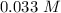

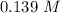
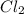 at 1000 K is= 0.033 M and the equilibrium concentration of
at 1000 K is= 0.033 M and the equilibrium concentration of 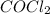 at 1000 K is 0.139 M
at 1000 K is 0.139 M 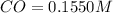
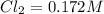
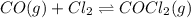
![K_c=\frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0519/6554/36d91.png)