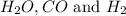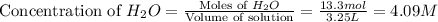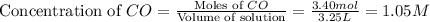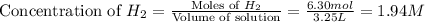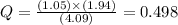The following reaction was carried out in a 3.25 L reaction vessel at 1100 K:
If during the course of the reaction, the vessel is found to contain 5.25 mol of C, 13.3 mol of H₂O, 3.40 mol of CO, and 6.30 mol of H₂, what is the reaction quotient Q?
Enter the reaction quotient numerically.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2


Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
The following reaction was carried out in a 3.25 L reaction vessel at 1100 K:
If during...
If during...
Questions



Mathematics, 19.03.2020 02:39


Biology, 19.03.2020 02:39






History, 19.03.2020 02:39

Business, 19.03.2020 02:39




Computers and Technology, 19.03.2020 02:39




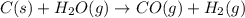
![Q=\frac{[CO][H_2]}{[H_2O]}](/tpl/images/0519/6055/f765a.png)