
Chemistry, 21.02.2020 17:42 macylen3900
Air bags are activated when a severe impact causes a steel ball to compress a spring and electrically ignite a detonator cap. This action causes sodium azide (NaN3) to decompose explosively according to the following reaction. 2 NaN3(s) → 2 Na(s) + 3 N2(g) What mass of NaN3(s) must be reacted to inflate an air bag to 70.6 L at STP?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3


Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3
You know the right answer?
Air bags are activated when a severe impact causes a steel ball to compress a spring and electricall...
Questions

Spanish, 23.04.2020 19:34


Mathematics, 23.04.2020 19:34




English, 23.04.2020 19:34


Mathematics, 23.04.2020 19:34

Biology, 23.04.2020 19:34


English, 23.04.2020 19:34



Biology, 23.04.2020 19:34




Biology, 23.04.2020 19:34

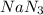 must be reacted to inflate an air bag to 70.6 L at STP.
must be reacted to inflate an air bag to 70.6 L at STP.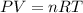
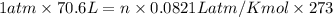
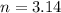
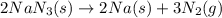
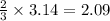 moles of
moles of 

