
Chemistry, 21.02.2020 02:27 nayelimoormann
Consider the following equilibrium process at 686°C. CO2(g) + H2(g) equilibrium reaction arrow CO(g) + H2O(g) The equilibrium concentrations of the reacting species are [CO] = 0.050 M, [H2] = 0.045 M, [CO2] = 0.086 M, and [H2O] = 0.040 M. (a) Calculate Kc for the reaction at 686°C. WebAssign will check your answer for the correct number of significant figures. (b) If we add CO2 to increase its concentration to 0.50 mol/L, what will the concentrations of all the gases be when equilibrium is reestablished?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
Consider the following equilibrium process at 686°C. CO2(g) + H2(g) equilibrium reaction arrow CO(g)...
Questions


Mathematics, 26.09.2021 02:30


Mathematics, 26.09.2021 02:30



Mathematics, 26.09.2021 02:30

Mathematics, 26.09.2021 02:40





Mathematics, 26.09.2021 02:40

English, 26.09.2021 02:40



Mathematics, 26.09.2021 02:40


Mathematics, 26.09.2021 02:40

Engineering, 26.09.2021 02:40

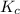 is 0.52.
is 0.52.![[CO_2] = 0.4748 M](/tpl/images/0518/6100/1098f.png)
![[H_2] = 0.0198 M](/tpl/images/0518/6100/4696c.png)
![[CO] = 0.0752 M](/tpl/images/0518/6100/1d075.png)
![[H_2O] =0.0652 M](/tpl/images/0518/6100/30296.png)
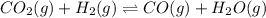
![[CO] = 0.050 M, [H_2] = 0.045 M, [CO_2] = 0.086 M, and [H_2O] = 0.040 M](/tpl/images/0518/6100/10b47.png)
![\K_c=\frac{[CO][H_2O]}{[CO_2][H_2]}](/tpl/images/0518/6100/2fcaf.png)
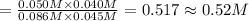
![K_c=\frac{[CO][H_2O]}{[CO_2][H_2]}](/tpl/images/0518/6100/c597d.png)
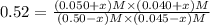
![[CO_2] = (0.50-x) M=(0.50-0.0252)M = 0.4748 M](/tpl/images/0518/6100/7357e.png)
![[H_2] = (0.045-x) M= (0.045-0.0252) M=0.0198 M](/tpl/images/0518/6100/31260.png)
![[CO] = (0.050+x) M=(0.050+0.0252)M = 0.0752 M](/tpl/images/0518/6100/0b9e4.png)
![[H_2O] = (0.040+x) M=(0.040+0.0252) M=0.0652 M](/tpl/images/0518/6100/b7e5e.png)


