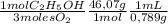Chemistry, 20.02.2020 08:36 ammullims822
The combustion of fuel in your car engine requires oxygen gas, which is supplied as air (21% oxygen molecules) into the engine. Consider a car that is using 100% ethanol, C2H5OH, as fuel. If your engine intakes 3.72 L of air per minute at 1.00 atm and 25ºC, what is the maximum volume of ethanol (0.789 g/mL) that can be burned per minute?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 02:30
In pea plants, the allele for tallness (t) is dominant to the allele for shortness (t). in the cross between a tall pea plant and a short pea plant shown below, what is the probability that the resulting offspring will be tall? whats the percent
Answers: 1

You know the right answer?
The combustion of fuel in your car engine requires oxygen gas, which is supplied as air (21% oxygen...
Questions

Biology, 31.01.2022 21:50

Mathematics, 31.01.2022 21:50



Mathematics, 31.01.2022 21:50

Mathematics, 31.01.2022 21:50

Mathematics, 31.01.2022 21:50



Mathematics, 31.01.2022 21:50


History, 31.01.2022 21:50

Chemistry, 31.01.2022 21:50




Chemistry, 31.01.2022 21:50


Mathematics, 31.01.2022 21:50