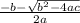Chemistry, 20.02.2020 08:37 thestuckone
At 700.°C, the total pressure of the system is found to be 3.01 atm. If the equilibrium constant KP is 1.52, calculate the equilibrium partial pressures of CO2 and CO.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1


Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1
You know the right answer?
At 700.°C, the total pressure of the system is found to be 3.01 atm. If the equilibrium constant KP...
Questions

Biology, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00

Physics, 10.03.2021 01:00

Geography, 10.03.2021 01:00


English, 10.03.2021 01:00

English, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00


Arts, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00

 = 1.5 atm
= 1.5 atm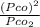
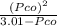 ⇒ 1.52 =
⇒ 1.52 = 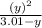 =
= 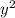 = 1.52 (3.01 - y)
= 1.52 (3.01 - y) 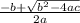 and y =
and y =