
Ethanol (C2H5OH) melts a - 144 oC and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kj/mol, and its enthalpy of vaporization is 38.56 kj/mol. The specific heats of solid and liquid ethanol are 0.97j/g - k and 2.3 j/g - K, respectively.
(a) How much heat is required to convert 42.0 g of ethanol at 35 °C to the vapor phase at 78 °C?
(b) How much heat is required to convert the same amount of ethanol at - 155 oC to the vapor phase at 78 °C?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
Ethanol (C2H5OH) melts a - 144 oC and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kj/m...
Questions


Biology, 05.11.2020 04:00

Mathematics, 05.11.2020 04:00

History, 05.11.2020 04:00



Mathematics, 05.11.2020 04:00

Mathematics, 05.11.2020 04:00


Mathematics, 05.11.2020 04:00


Mathematics, 05.11.2020 04:00




Mathematics, 05.11.2020 04:00

History, 05.11.2020 04:00



Mathematics, 05.11.2020 04:00

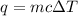 .........(1)
.........(1) = specific heat capacity of substance
= specific heat capacity of substance = change in temperature
= change in temperature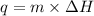 ........(2)
........(2)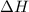 = enthalpy of the reaction
= enthalpy of the reaction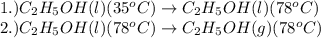
![m=42.0g\\c_l=2.3J/g.K\\T_2=78^oC\\T_1=35^oC\\\Delta T=[T_2-T_1]=[78-35]^oC=43^oC=43K](/tpl/images/0516/7715/feb03.png)
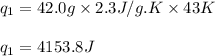
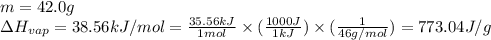
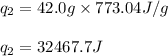
![[q_1+q_2]](/tpl/images/0516/7715/7bba2.png)
![[4153.8J+32467.7J]=36621.5J](/tpl/images/0516/7715/59544.png)
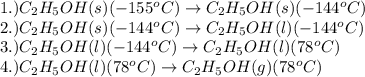
![m=42.0g\\c_s=0.97J/g.K\\T_2=-144^oC\\T_1=-155^oC\\\Delta T=[T_2-T_1]=[-144-(-155)]^oC=11^oC=11K](/tpl/images/0516/7715/d4732.png)
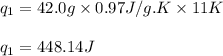
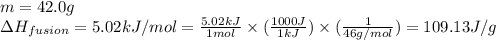
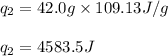
![m=42.0g\\c_l=2.3J/g.K\\T_2=78^oC\\T_1=-144^oC\\\Delta T=[T_2-T_1]=[78-(-144)]^oC=222^oC=222K](/tpl/images/0516/7715/36530.png)
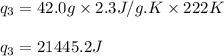
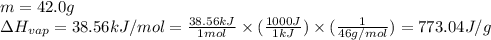
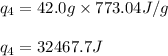
![[q_1+q_2+q_3+q_4]](/tpl/images/0516/7715/4ee42.png)
![[448.14+4583.5+21445.2+32467.7]J=58944.5J](/tpl/images/0516/7715/a8249.png)


