
Chemistry, 19.02.2020 22:59 Picklehead1166
Sodium metal (atomic weight 22.99 g/cm^3) adopts a body-centered cubic structure with a density of 0.97 g/cm^3. (a) Use this information and Avogrado's number (Na=6.022x10^23) to estimate the atomic radius of sodium. (b) If it didn't react so vigorously, sodium could float on water. Use the answer from part (a) to estimate the density of Na if its structure were that of a cubic close-packed metal. Would it still float on the water?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 12:30
The missing component to the table to the right or indicated with orange letters complete the table by filling in the corresponding numbers or symbols
Answers: 3

You know the right answer?
Sodium metal (atomic weight 22.99 g/cm^3) adopts a body-centered cubic structure with a density of 0...
Questions

Biology, 30.11.2020 19:30


Mathematics, 30.11.2020 19:30

Arts, 30.11.2020 19:30


Biology, 30.11.2020 19:30


Mathematics, 30.11.2020 19:30



Health, 30.11.2020 19:30

Computers and Technology, 30.11.2020 19:30


Health, 30.11.2020 19:30





English, 30.11.2020 19:30

Mathematics, 30.11.2020 19:30

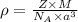
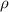 = density =
= density = 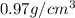
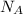 = Avogadro's number =
= Avogadro's number = 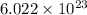
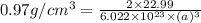
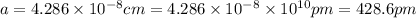
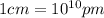
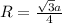
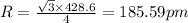
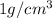
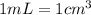 )
)

