
Chemistry, 19.02.2020 06:01 insulation62
Learning Goal: To become familiar with the octet rule and to use it to determine the charges of ions. Noble gases are especially stable because their valence shells are full. This is referred to as having an octet, because most valence shells contain eight electrons. Other elements will gain or lose electrons to achieve an octet and become an ion. A positively charged ion has lost electrons and is called a cation whereas a negatively charged ion has gained electrons and is called an anion. Nonmetals tend to gain electrons to form anions Metals tend to lose electrons to form cations.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 14:30
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2
You know the right answer?
Learning Goal: To become familiar with the octet rule and to use it to determine the charges of ions...
Questions


History, 11.09.2019 19:10



History, 11.09.2019 19:10

Mathematics, 11.09.2019 19:10

Mathematics, 11.09.2019 19:10




English, 11.09.2019 19:10

Mathematics, 11.09.2019 19:10

Health, 11.09.2019 19:10

Mathematics, 11.09.2019 19:20

Mathematics, 11.09.2019 19:20



Physics, 11.09.2019 19:20


Mathematics, 11.09.2019 19:20

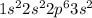 .
. 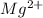 ion.
ion.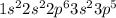 . To attain noble gas configuration it will readily accept an electron from a donor atom.
. To attain noble gas configuration it will readily accept an electron from a donor atom.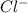 ion.
ion.

