
Chemistry, 19.02.2020 05:52 animerocks07
Determining reaction order : Rate Laws(Chemistry)
The reaction of nitric oxide with hydrogen at 1280C is as follows:
2NO(g) +2H2 (g) -> N2(g) + 2H2O(g)
From the following experimental data, determine the rate law and the rate constant.
30 POINTS!


Answers: 2
Another question on Chemistry



Chemistry, 23.06.2019 01:00
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1

Chemistry, 23.06.2019 09:30
If the solubility of a gas in water is 1.22g/2.75 atm, what is it’s solubility (in g/l) at 1.0 atm
Answers: 1
You know the right answer?
Determining reaction order : Rate Laws(Chemistry)
The reaction of nitric oxide with hydrogen...
The reaction of nitric oxide with hydrogen...
Questions


Mathematics, 20.06.2021 06:30

Biology, 20.06.2021 06:30


English, 20.06.2021 06:30


Mathematics, 20.06.2021 06:40

Mathematics, 20.06.2021 06:40


Computers and Technology, 20.06.2021 06:40

Mathematics, 20.06.2021 06:40

History, 20.06.2021 06:40

English, 20.06.2021 06:40

Business, 20.06.2021 06:40


Mathematics, 20.06.2021 06:40



Physics, 20.06.2021 06:40

Mathematics, 20.06.2021 06:40

![R=k[NO]3[H_2]^1](/tpl/images/0515/4935/da8f4.png)
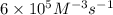
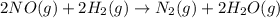
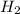 in rate law be x and y .
in rate law be x and y .![R=k[NO]^[H_2]^y](/tpl/images/0515/4935/cf38a.png)
![[NO]=0.0100 M, [H_2]=0.0100 M](/tpl/images/0515/4935/aa668.png)
![0.00600 M/s=k[0.0100 M]^x[0.0100 M]^y](/tpl/images/0515/4935/a6007.png) ..[1]
..[1]![[NO]=0.0200 M, [H_2]=0.0300 M](/tpl/images/0515/4935/ba703.png)
![0.144 M/s=k[0.0200 M]^x[0.0300 M]^y](/tpl/images/0515/4935/0042d.png) ..[2]
..[2]![[NO]=0.0100 M, [H_2]=0.0200 M](/tpl/images/0515/4935/ba63c.png)
![0.0120 M/s=k[0.0100 M]^x[0.0200 M]^y](/tpl/images/0515/4935/9f0d9.png) ..[3]
..[3]![\frac{0.00600 M/s}{0.0120 M/s}=\frac{k[0.0100 M]^x\times [0.0100 M]^y}{k[0.0100 M]^x\times [0.0200 M]^y}](/tpl/images/0515/4935/1b196.png)
![\frac{0.00600 M/s}{0.144 M/s}=\frac{k[0.0100 M]^x\times [0.0100 M]^1}{k[0.0200 M]^x\times [0.0300 M]^1}](/tpl/images/0515/4935/e9761.png)
![0.00600 M/s=k[0.0100 M]^3[0.0100 M]^1](/tpl/images/0515/4935/2cbdd.png) ..[1]
..[1]![k=\frac{0.00600 M/s}{[0.0100 M]^3[0.0100 M]^1}=6\times 10^5 M^{-3}s^{-1}](/tpl/images/0515/4935/88d8d.png)


