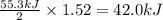A chemist measures the energy change during the following reaction: 1. This reaction is:. a. endothermic. b. exothermic. 2. Suppose 70.1 g of NO2 react. Will any heat be released or absorbed? A. Yes, absorbed. B. Yes, released. C. No. 3. If you said heat will be released or absorbed, calculate how much heat will be released or absorbed?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1


You know the right answer?
A chemist measures the energy change during the following reaction: 1. This reaction is:. a. endothe...
Questions






Mathematics, 03.10.2019 07:00






History, 03.10.2019 07:00

Physics, 03.10.2019 07:00


Mathematics, 03.10.2019 07:00

Mathematics, 03.10.2019 07:00

History, 03.10.2019 07:00

Mathematics, 03.10.2019 07:00


History, 03.10.2019 07:00

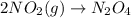
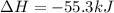 1. This reaction is:______. a. endothermic. b. exothermic.
1. This reaction is:______. a. endothermic. b. exothermic. 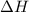 for Endothermic reaction is positive and
for Endothermic reaction is positive and 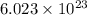 of particles.
of particles.
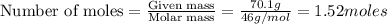
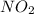 reacts, energy released = 55.3 kJ
reacts, energy released = 55.3 kJ