
The rate law for the reaction
2NO(g) + Cl2(g) → 2NOCl(g)
is given by R = k[NO][Cl2]
If the following is the mechanism for the reaction, NO(g) + Cl2(g) → NOCl2(g) NOCl2(g) + NO(g) → 2NOCl(g)
which of the following statements accurately describes this reaction? Check all that apply.
-2nd order reaction
-The first step is the slow step.
-Doubling [NO] would quadruple the rate.
-Cutting [Cl2] in half would decrease the rate by a factor of two.
-The molecularity of the first step is 1.
-Both steps are termolecular.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
The wave shown on the electromagnetic spectrum disturb the medium it passes through a)different frequency. b)the same frequency .
Answers: 2

Chemistry, 22.06.2019 07:00
The blackbody curve for a star name zeta is shown below. what is the peak wavelength for this star ?
Answers: 1

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
The rate law for the reaction
2NO(g) + Cl2(g) → 2NOCl(g)
is given by R = k...
2NO(g) + Cl2(g) → 2NOCl(g)
is given by R = k...
Questions





Computers and Technology, 25.01.2020 00:31

Mathematics, 25.01.2020 00:31

Mathematics, 25.01.2020 00:31


Mathematics, 25.01.2020 00:31




History, 25.01.2020 00:31


English, 25.01.2020 00:31





Chemistry, 25.01.2020 00:31

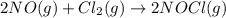
![k[NO][Cl_{2}]](/tpl/images/0514/1761/8cd0b.png)
 . Since, power of the concentrations of each of these is equal to 1. Therefore, order of the reaction is equal to 1 + 1 = 2.
. Since, power of the concentrations of each of these is equal to 1. Therefore, order of the reaction is equal to 1 + 1 = 2.

